Rotec Tablet Uses In Urdu | Rotec 75 Tablet Uses In Urdu | Rotec Tablet 50 Mg Uses In Urdu | Rotec - YouTube

Rotec Tablet Uses In Urdu | Rotec 75 Tablet Uses In Urdu | Rotec Tablet 50 Mg Uses In Urdu | Rotec - YouTube
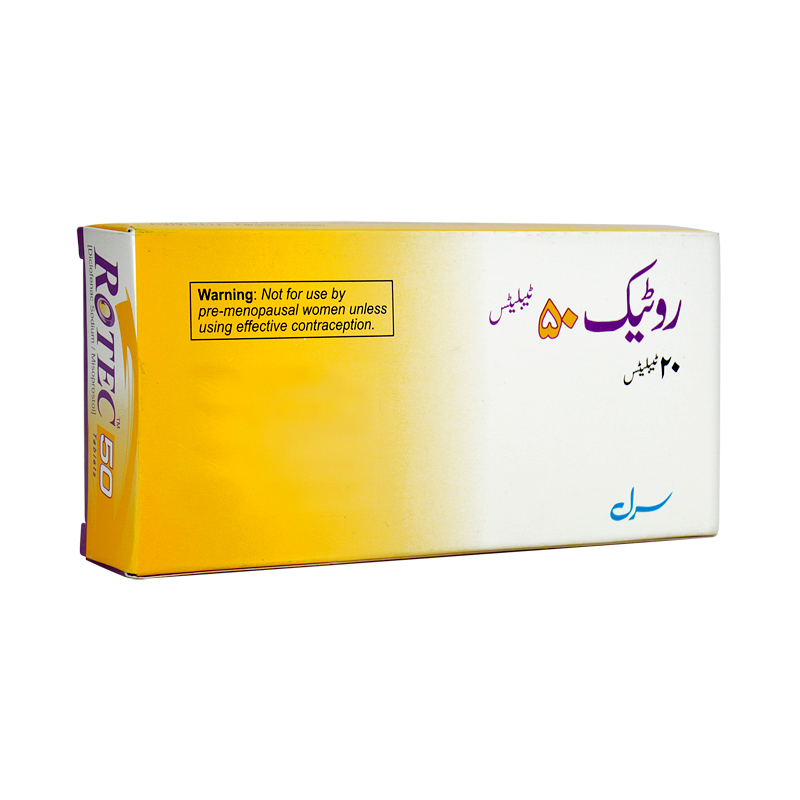
![FDA Advisory No.2023-0210 || 1. Public Health Warning Against the Purchase and Use of the Unregistered Drug Product “SEARLE ROTEC™50 Tablets [Diclofenac Sodium/ Misoprostol]” - Food and Drug Administration FDA Advisory No.2023-0210 || 1. Public Health Warning Against the Purchase and Use of the Unregistered Drug Product “SEARLE ROTEC™50 Tablets [Diclofenac Sodium/ Misoprostol]” - Food and Drug Administration](https://www.fda.gov.ph/wp-content/uploads/2023/02/FDA-Advisory-No.2023-0210.png)
FDA Advisory No.2023-0210 || 1. Public Health Warning Against the Purchase and Use of the Unregistered Drug Product “SEARLE ROTEC™50 Tablets [Diclofenac Sodium/ Misoprostol]” - Food and Drug Administration

herbsmedicine2013 on X: "100% LEGIT Transaction TRUSTED SELLER SINCE 2013 CASH ON DELIVERY! 09363314860 fb Page: https://t.co/GJJDghZTXY ☑️Door to door delivery via LBC (secure and safe) ☑️Results Within 24 hours #MANILA #PAMPANGA #















.jpg)